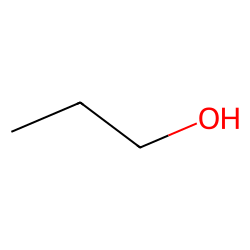
Molecular Weight Dependence of Block Copolymer Micelle Fragmentation Kinetics | Journal of the American Chemical Society
Hydrogen | Free Full-Text | Energetics of LOHC: Structure-Property Relationships from Network of Thermochemical Experiments and in Silico Methods
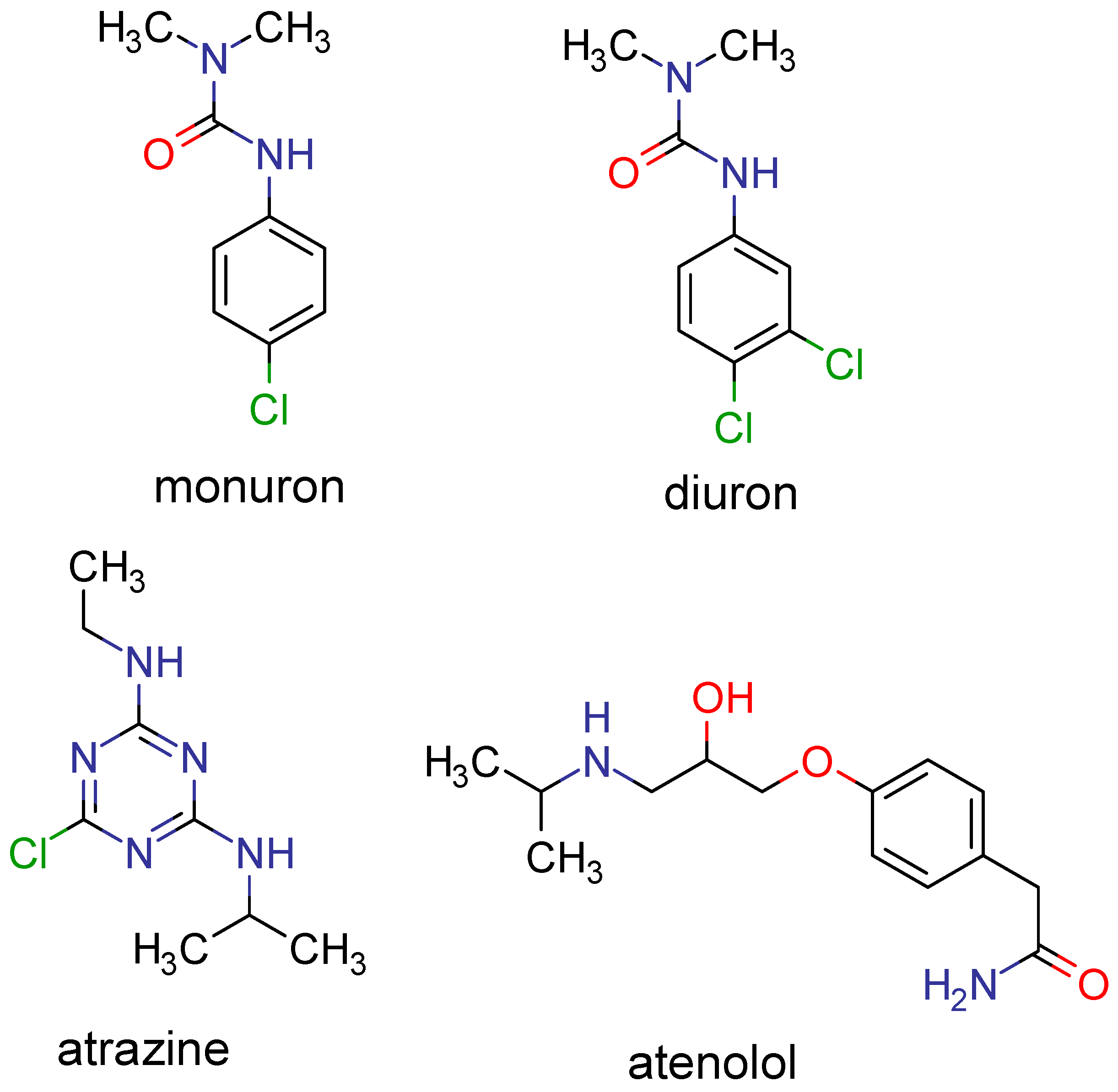
Processes | Free Full-Text | Predicting the Solubility of Nonelectrolyte Solids Using a Combination of Molecular Simulation with the Solubility Parameter Method MOSCED: Application to the Wastewater Contaminants Monuron, Diuron, Atrazine and

Energies | Free Full-Text | Insight into the Thermodynamic Properties of Promising Energetic HNTO·AN Co-Crystal: Heat Capacity, Combustion Energy, and Formation Enthalpy
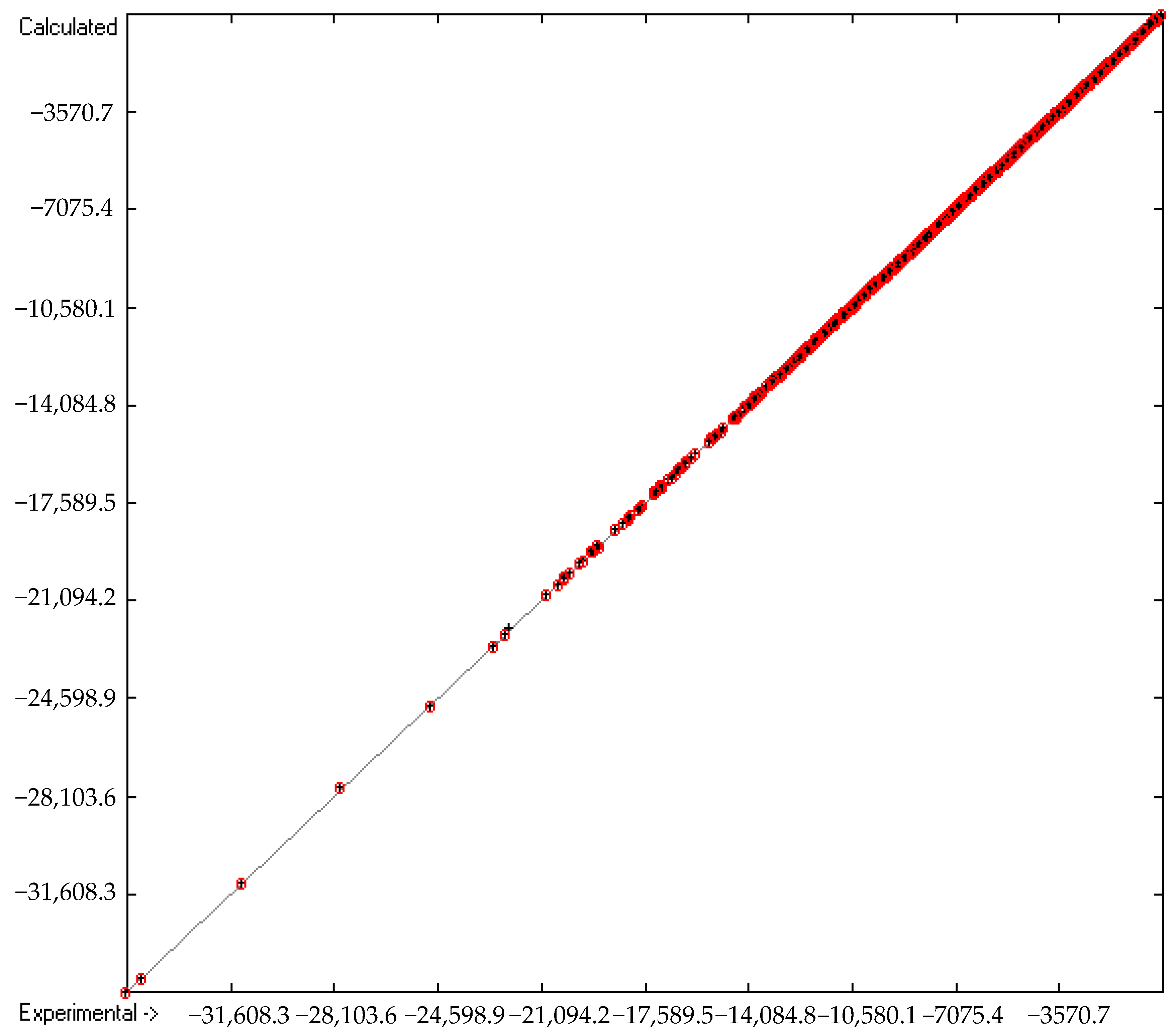
Molecules | Free Full-Text | Revision and Extension of a Generally Applicable Group-Additivity Method for the Calculation of the Standard Heat of Combustion and Formation of Organic Molecules

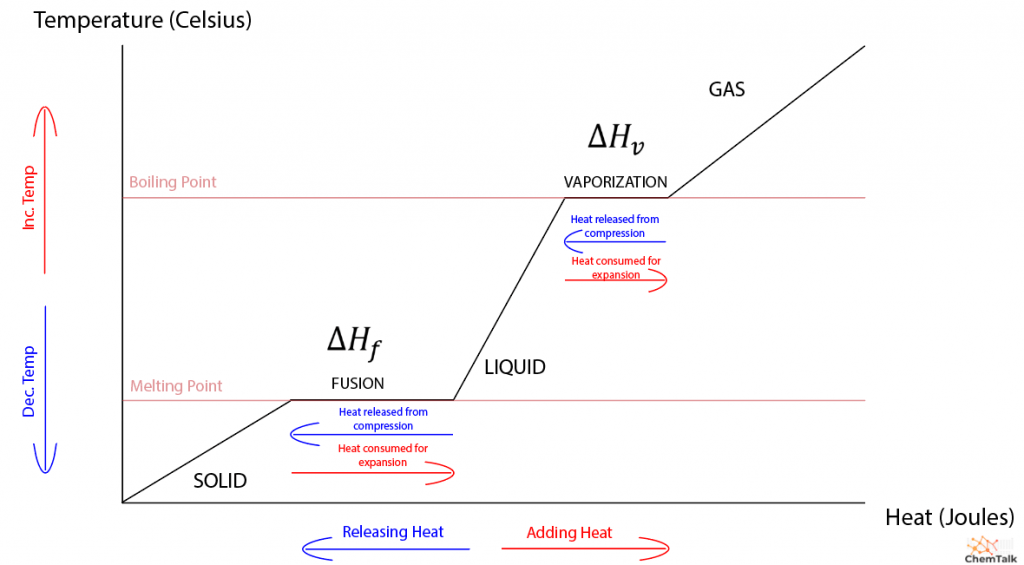
Define enthalpy of sublimation. How is it related to enthalpy of fusion and enthalpy of vaporization?
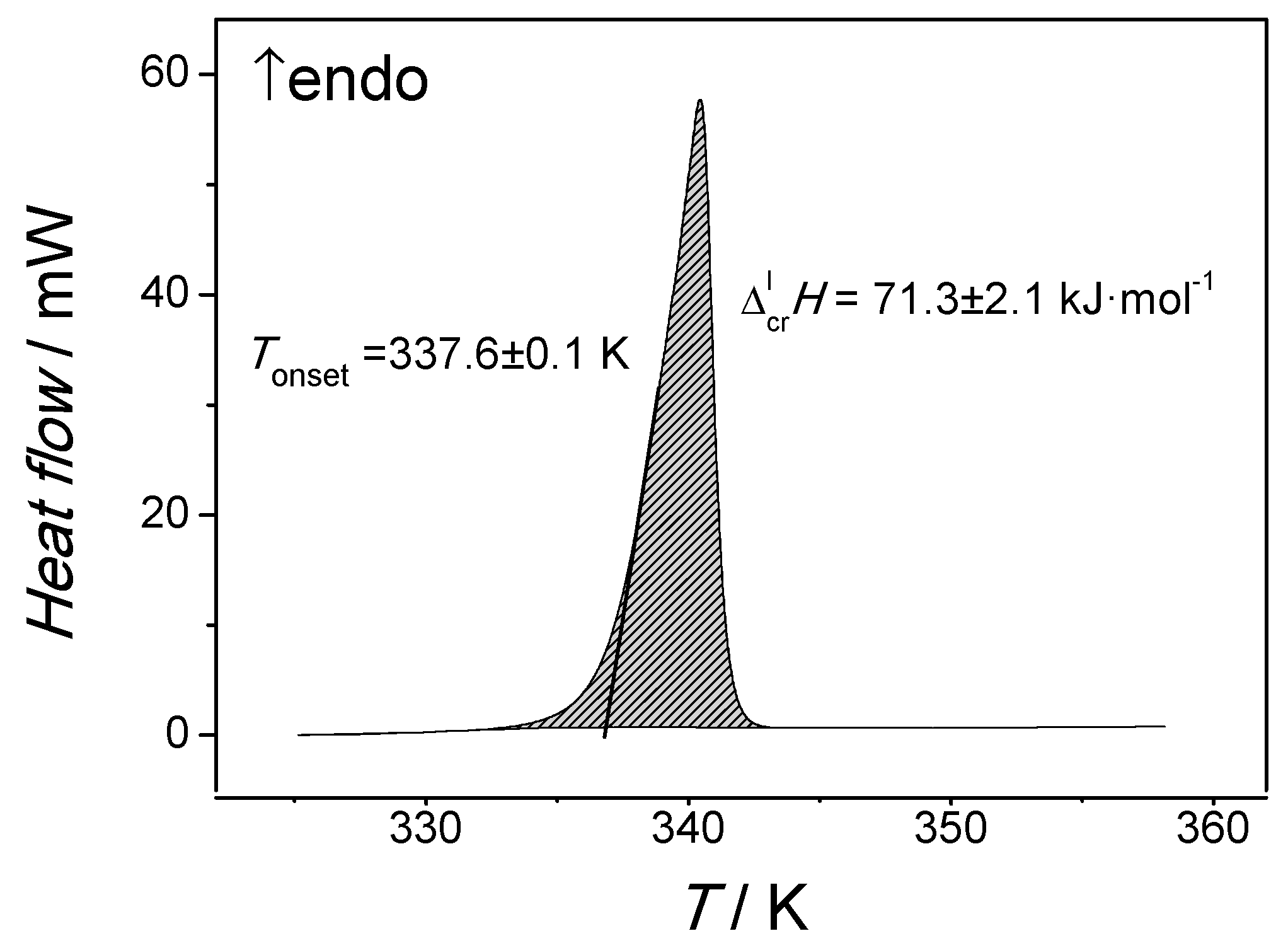
Liquids | Free Full-Text | Application of Solution Calorimetry to Determining the Fusion Enthalpy of an Arylaliphatic Compound at 298.15 K: n-Octadecanophenone
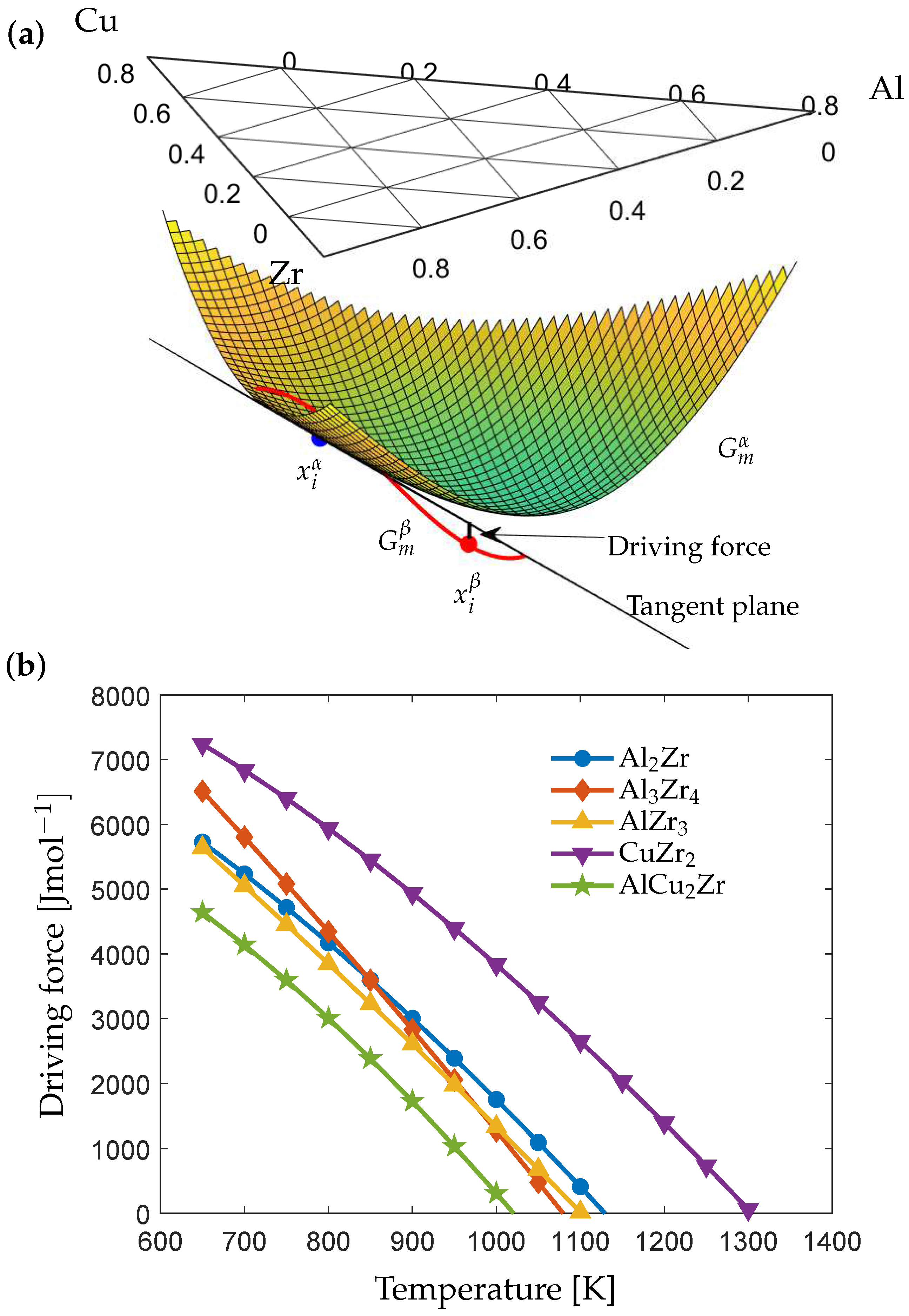
Metals | Free Full-Text | Modeling of Diffusion-Controlled Crystallization Kinetics in Al-Cu-Zr Metallic Glass

Evaluation of the Gibbs Free Energy Changes and Melting Temperatures of DNA/DNA Duplexes Using Hybridization Enthalpy Calculated by Molecular Dynamics Simulation | The Journal of Physical Chemistry B

The heat of fusion, melting temperature of KNO3, NaNO3, Solar salt and... | Download Scientific Diagram

Active Thermochemical Tables: Enthalpies of Formation of Bromo- and Iodo-Methanes, Ethenes and Ethynes | The Journal of Physical Chemistry A

Enthalpy changes (ΔH in J g −1 ) associated with fusion (melting) of... | Download Scientific Diagram